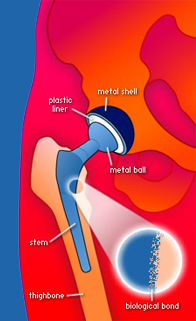
Total hip prosthesis
- Replacement of worn surfaces
- Metal socket with plastic liner
- Metal ball on a metal stem
- Cemented or uncemented
- Other bearing surfaces designs
- Ceramic-on-ceramic
- Metal-on-metal
The goal of total hip replacement is to remove the worn surfaces of the arthritic joint and replace them with new, artificial surfaces. In the case of total hip, the artificial socket is made of an outer metal shell and an inner plastic liner. Occasionally the surgeon may choose to insert an all-plastic socket.
On the femoral (thigh bone) side a metal ball attached to a metal stem, which is then inserted into the thighbone.
The prosthesis can be attached to the bone with help of bone cement, or without cement. The uncemented prosthesis initially relies on tight fit. Later on, the bone will grow into the porous surface of the prosthesis and form a biological bond. In my practice, I almost exclusively use the uncemented total hips.
There are some other designs, which may prolong the longevity of the prosthesis. Since the most common cause of long-tem failure is related to wear of the plastic liner, a different type of bearing surfaces (ceramic-on-ceramic) is occasionally used. The operation itself is otherwise the same. The only difference is that at a particular time during the operation rather than inserting a plastic liner, one inserts a ceramic part, and instead of a metal ball, one would insert a ceramic one. There is no proof that these types of prosthesis will last longer. There is, however, simulation laboratory data and early clinical results to consider this design in the younger patients (under 55-60 years). For the patients above 60 it is probably safer to go with the standard prosthesis proven to work well for many years.
The other type of bearing surfaces that used to be considered to be superior to the standard metal-on-plastic was metal-on-metal. Hundreds of thousand of such prostheses were implanted with the hopes of solving the “premature” wear problem. Unfortunately such “metal-on-metal” implants turned out not to hold promise and failed early in higher numbers than expected. The majority of manufacturers have pulled this type of implant from the market.